S2 Molecular Orbital Diagram
Involved intense orbitals relatively Molecular orbital theory Molecular orbitals bond order bonding electrons chemistry ion has unpaired geometry delocalized chemwiki exercises structure answers general principles v1 covalent
S-orbital | physics | Britannica
Inorganic chemistry Orbital diagrams and electron configuration How does the 3s orbital differ from the 2s orbital?
Figure s2. the molecular orbitals involved in the relatively intense
8.3 development of quantum theory – chem 1114 – introduction to chemistryNeed a molecular orbital diagram for s2, s2+ and s2- not the Orbital study calculationSolved consider the partially filled-in molecular orbital.
Why is o2 paramagnetic?Chapter 6.5 delocalized bonding and molecular orbitals Molecular orbital theoryContributing molecular transitions orbitals pristine.

Orbital molecular theory n2 orbitals diatomic valence o2 atomic carbon homonuclear sp3 molecule majors cnx chem atoms
Draw the valence shell molecular orbital diagram of the oxygen moleculeChapter 6.5 delocalized bonding and molecular orbitals Orbital electron diagrams configuration chemistry practice problems basicO2 molecular orbital diagrams.
Orbital orbitals electron atoms science chemistry britannicaBond order molecular orbital theory magnetic chemistry properties strength Molecular orbitals contributing to (a) s0 → s1 and s0 → s2 transitionsMolecular orbital theory.

Orbital cl2 orbitals bonding delocalized atoms energies chem chapter general libretexts chemistry formed adjacent
Orbital valence mot chemistry electrons draw moleculeOrbital 2s 3s differ does socratic orbitals Orbital molecular molecules diagram orbitals diatomic bonding of2 delocalized bond atomic libretexts electrons chem correlation hybridization atoms np homonuclear pageindexMolecular orbital electrons 3s 3p partially filled solved.
Mo o2 orbital molecular theory orbitals paramagnetic bond diagram oxygen order why configuration atomic electrons energy unpaired diagrams two loneFigure s2. the molecular orbitals involved in the relatively intense Intense relatively orbitals molecularChemistry 101: molecular orbital theory, bond order, bond strength.

Orbital molecular diagram cl2 s2 molecule mot unpaired orbitals bond electron bonding draw molecules c2 mo energy theory valence electrons
Orbital molecular diagram bn mo orbitals bond diagrams order cl2 theory paramagnetic energy level draw bonding valence electrons chemistry homonuclearThe orbitron: 3s atomic orbitals Orbital atomic 3s orbitals 6s orbitron shefOrbital o2 oxygen theory paramagnetic diagrams ozone chemistry molecule bonding libretexts valence molecules orbitals predicts chem electrons.
Chem orbitals shapes quantum chemistry model atoms theory orbital diagram electrons sublevels sublevel mechanics using wave axes spherical figure shaped .
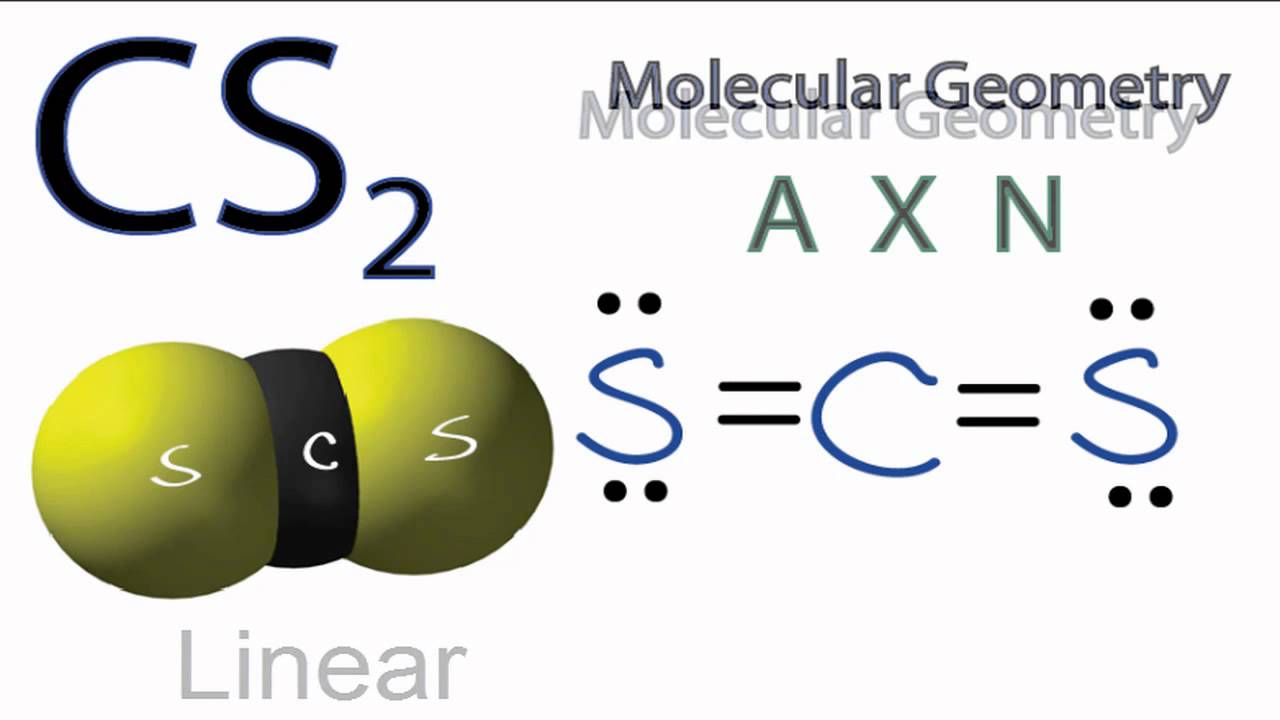

...

...

...

...

8.3 Development of Quantum Theory – CHEM 1114 – Introduction to Chemistry

...

Molecular Orbital Theory - Chemistry LibreTexts

How does the 3s orbital differ from the 2s orbital? | Socratic